lamivudine salicylate
lamivudine salicylate
CAS: 173522-96-8
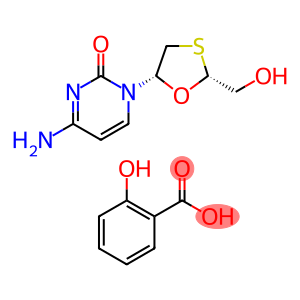
Molecular Formula: C8H11N3O3S.C7H6O3
lamivudine salicylate - Names and Identifiers
| Name | lamivudine salicylate |
| Synonyms | lamivudine salicylate 2r-(Hydroxymethyl)-5(S)-Cytosin-1-Yl-1,3-Oxathiolanesalicycic Acid Salt 2R-(HYDROXYMETHYL)-5(S)-CYTOSIN-1-YL-[1,3]-OXATHIOLANE SALICYLIC ACID SALT (2R, 5S) -2-(Hydroxymethyl)-5-(4-amino-2-oxo-1(2H)-pyrimidinyl)-1,3-oxathiolane salicylate (2r-cis)-4-amino-1-[2-(hydroxymethyl)-1,3-oxathiolan-5-yl]-2(1h)-pyrimidinone 2-hydroxybenzoate 2R-(HYDROXYMETHYL)-5(S)-CYTOSIN-1-YL-1,3-OXATHIOLANESALICYCIC ACID SALT (LAMIVUDINE SALICYLATE) 2R-(hydroxymethyl)-5(S)-cytosin-lyl-1,3-oxothiolane salicylic acid salt (Lamivudine Salicylate) 2R-(hydroxymethyl)-5(s)-cytosin-1-yl-[1,3]-oxathiolane salicylic acid salt (intermediate of lamivudine) |
| CAS | 173522-96-8 |
| EINECS | 827-409-4 |
| InChI | InChI=1/C8H11N3O3S.C7H6O3/c9-5-1-2-11(8(13)10-5)6-4-15-7(3-12)14-6;8-6-4-2-1-3-5(6)7(9)10/h1-2,6-7,12H,3-4H2,(H2,9,10,13);1-4,8H,(H,9,10)/t6-,7+;/m1./s1 |
lamivudine salicylate - Physico-chemical Properties
| Molecular Formula | C8H11N3O3S.C7H6O3 |
| Molar Mass | 367.38 |
| Storage Condition | 2-8°C |
lamivudine salicylate - Reference Information
| overview | lamivudine is a pharmaceutical raw material with good antiviral activity. it is mainly used for anti-hepatitis B and anti-HIV virus. it is the main active ingredient in the current cocktail therapy for treating hepatitis B and AIDS. its synthesis needs to be excessive through the synthesis of intermediates. Lamivudine intermediate HDMS lamivudine salicylic acid lamivudine molecular formula: C15H17O6N3S.H2O, its chemical name: (2R-cis)-4-amino -1-(2-hydroxymethyl-1, 3-oxothiacycl-5-yl)-1H-pyrimidin-2-ketosalicylate. During the synthesis of lamivudine intermediates, it is reported publicly at home and abroad. The route published by the U.S. patent is based on 2-bromodiethoxyethane as the raw material, and sodium hydroxide is hydrolyzed after reacting with thiobenzoic acid to obtain 2-Ryukyu diethoxyethane, which is reacted with benzoyloxyacetaldehyde to obtain 2-benzoyloxymethyl-5-ethoxyl-1, 3-oxothelane pentane, and then catalyzed with trimethylsilane trifluoromethanesulfonate, and cytosine protected with trimethylsilane Reaction, acetylation, column chromatography to remove trans, the cis-product is obtained after deacetylation; the acetylated cis-product reacts with menthol chloroformate, recrystallizes to separate (2R,5S) isomers, and ammonolysis to obtain lamivudine. This method 2-Ryukyu diethoxyethane reacts with benzoyloxyacetaldehyde to obtain 2-benzoyloxymethyl-5-ethoxyl-1, 3-oxothalane. The reaction yield is low and the raw material is complex, it is also necessary to remove the trans body, resulting in a greatly reduced yield, increased cost, and the use of chromatography columns is not conducive to industrial production. |
Last Update:2024-04-09 20:49:11
Supplier List
Product Name: lamivudine salicylate Request for quotation
CAS: 173522-96-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 173522-96-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Lamivudine salicylate Request for quotation
CAS: 173522-96-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 173522-96-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Lamivudine salicylate Visit Supplier Webpage Request for quotation
CAS: 173522-96-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 173522-96-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Product Name: lamivudine salicylate Request for quotation
CAS: 173522-96-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 173522-96-8
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: Lamivudine salicylate Request for quotation
CAS: 173522-96-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 173522-96-8
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: Lamivudine salicylate Visit Supplier Webpage Request for quotation
CAS: 173522-96-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 173522-96-8
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


